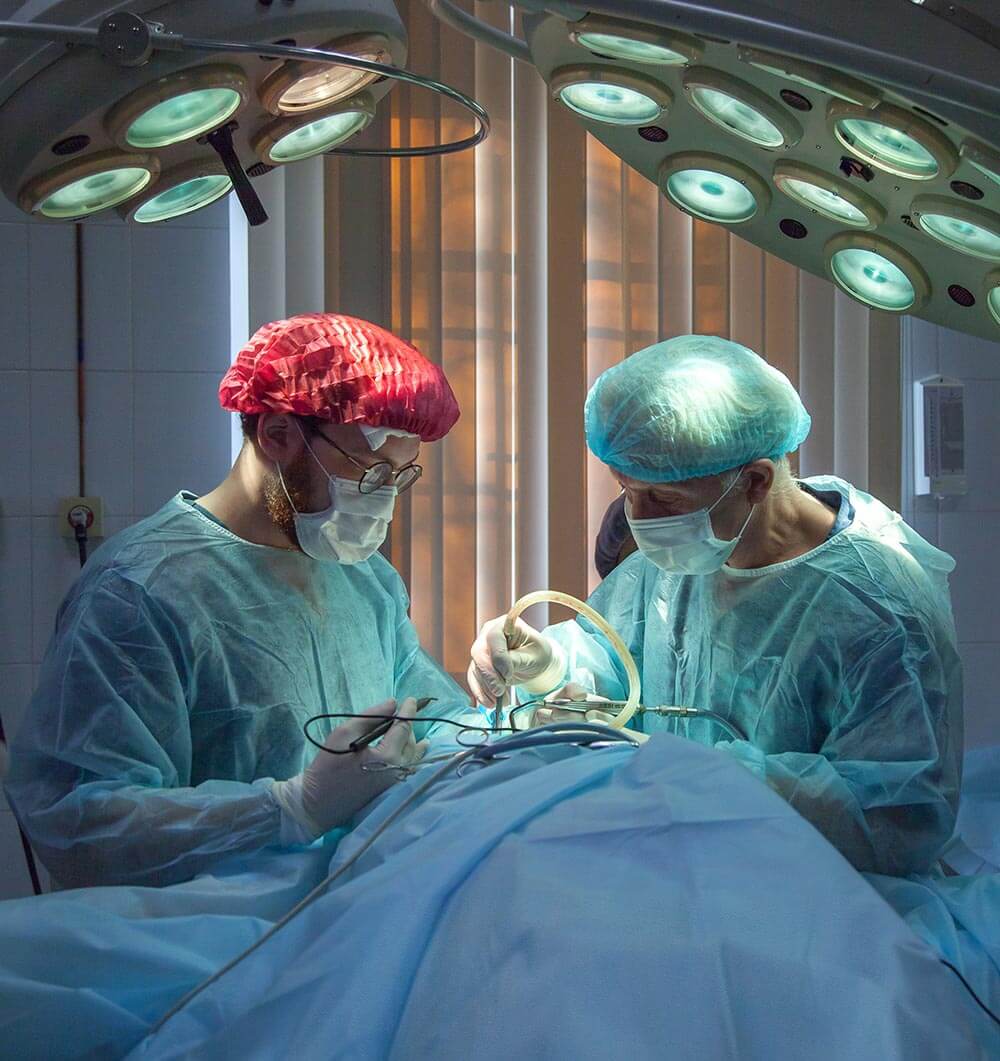


ISO 13485 is an international standard that specifies requirements for a quality management system where an organization needs to demonstrate its ability to provide medical devices and related services that consistently meet customer and applicable regulatory requirements.
ISO 13485 is a standard intended primarily for organizations that are involved in one or more stages of the life cycle, including design and development, manufacturing, storage and distribution, installation, or servicing of a medical device.

Potrebujete poradiť ohľadne ISO 13485 Systém riadenia kvality zdravotníckych prostriedkov? Neváhajte nás kontaktovať na telefónnom čísle +421 905 325 399, alebo vyplňte krátky formulár nižšie a my vám zavoláme.
Do you have any questions about our services or are you interested in a quote? Leave us your contact details and we will get back to you as soon as possible.